Advancing AI-Driven Drug Discovery: A Data-Centric Perspective from a:head bio
In the evolving landscape of drug discovery, artificial intelligence is often framed as the headline innovation. Yet, as Josh Bagley, CSO of a:head bio, makes clear, the real differentiator lies not just in algorithms—but in the quality and relevance of the underlying biological data.
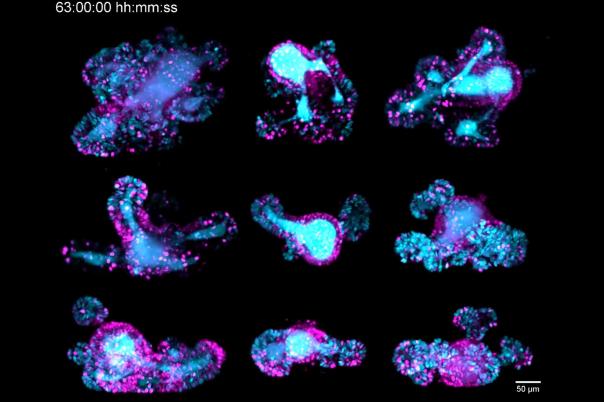
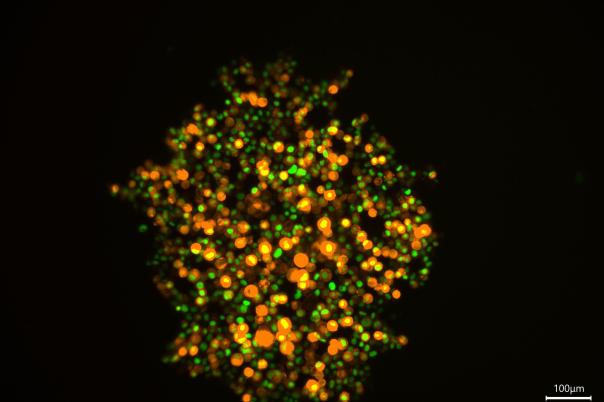
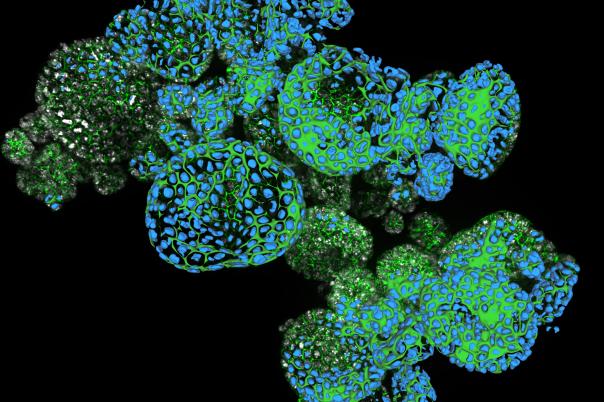
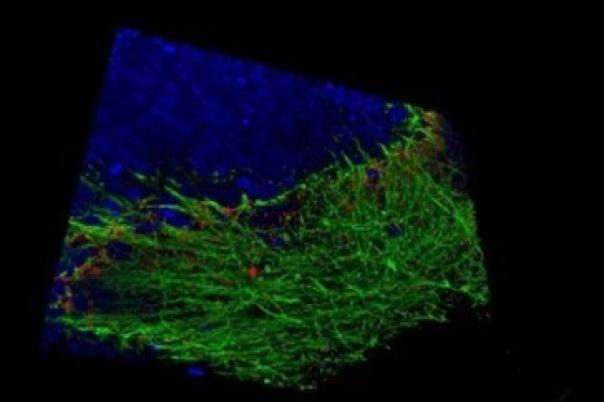
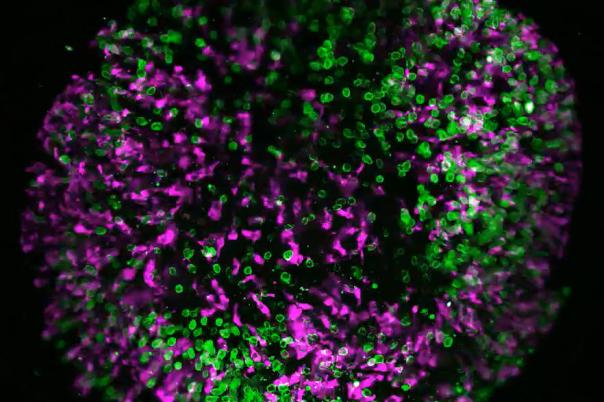
a:head bio, based in Vienna, is taking a distinctly human biology-first approach. The company leverages human brain organoids—lab-grown tissue models that mimic aspects of the human brain—to generate high-resolution datasets tailored to neurological disorders. These organoids form the backbone of a platform designed to improve drug screening and development across the pipeline.
AI as an Enabler, Not the Core Asset
Bagley is pragmatic about AI’s role. Rather than positioning a:head bio as an “AI company,” he describes AI as a critical enabler—particularly in neuroscience, where complexity has historically limited interpretation. AI excels at identifying patterns in dense, multidimensional datasets, helping researchers extract meaning from biological signals that were previously inaccessible.
However, AI’s effectiveness is directly tied to data quality. “You still need to get into the lab and produce the data,” Bagley notes, emphasizing that despite technological advances, high-resolution human data remains sparse. This data gap continues to be a bottleneck in translating computational insights into viable therapies.
The Power of Unique Human Data
a:head bio’s competitive edge lies in its proprietary organoid-derived datasets. These provide functional, human-relevant validation that complements other data sources such as patient data, chemical binding profiles, and multi-omics. When integrated, these datasets have the potential to uncover novel targets and disease pathways—pushing drug discovery beyond traditional boundaries.
While fully in silico drug development remains aspirational, Bagley a hybrid future where biological systems like organoids play a critical validation role alongside computational models.
Scaling the Biology
A key milestone for a:head bio is scalability. Historically, organoid production has been low-throughput and time-intensive. The company has focused on developing scalable protocols, with the next 12–18 months aimed at enabling high-throughput screening of complex brain tissues. This shift could significantly accelerate early-stage drug discovery.
Collaboration as a Growth Strategy
For a specialized biotech like a:head bio, partnerships are not optional—they are essential. Collaborations with device manufacturers enable advanced readouts of organoid activity, while partnerships with pharmaceutical companies are critical for clinical translation. In an increasingly interdisciplinary field, no single organization can cover the full spectrum of expertise.
Looking ahead: Reusable Data and Better Outcomes
Looking to the future, Bagley is particularly optimistic about data reusability. Traditionally, much of the data generated in drug screening—especially negative results—has been underutilized. AI now offers the to aggregate, reinterpret, and reuse these datasets across contexts, compounding their value over time.
This shift toward reusable, cumulative data ecosystems could mark a turning point in drug development efficiency—reducing redundancy, lowering costs, and ultimately improving patient outcomes.
Conclusion
a:head bio’s approach highlights a critical insight for the industry: AI alone is not a silver bullet. Its true potential is unlocked when paired with rich, human-relevant data. As organoid technologies scale and datasets grow, the convergence of biology and computation may finally deliver on the long-standing promise of more predictive, efficient drug discovery.